the Project
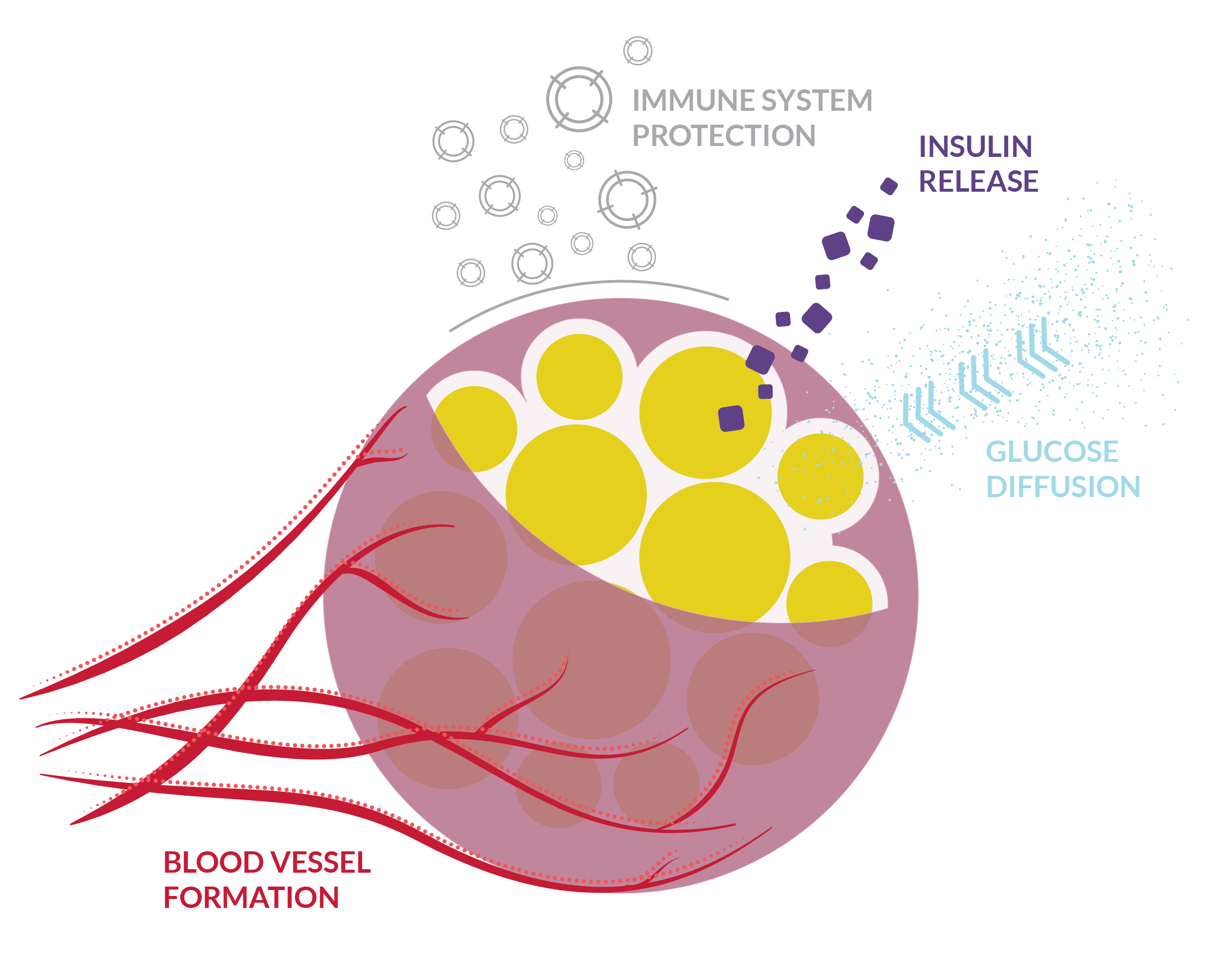
an encapsulating strategy based on a bio-inspired material that mimics elastin

stay connected with us
We are an international, multidisciplinary team of scientists from different fields ranging from synthetic biology to nanobiotechnology, molecular and cellular biology.
Our consortium gathers 12 Partners spread across 8 countries including Universities, SMEs and research institutes.
Our project is funded under the European Commission’s Framework Programme for Research, Innovation and Technology Horizon2020.
Since June 2015, we are cooperating with a cross-disciplinary strategy and a unique objective:
to develop a smarter strategy to cope with diabetes.
Prof. José Carlos Rodríguez Cabello